Age-based approvals for rare diseases can be devastating
For most of my life, my family advocated for therapies to help my twin brother. Although the recent approval of a new drug should give us reason to celebrate, there is still work to be done.
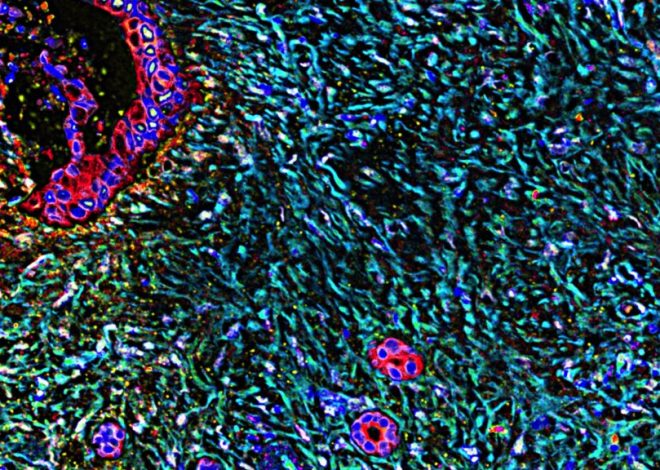
My twin brother is 28 years old and suffers from severe Hunter syndrome, also known as mucopolysaccharidosis (MPS) type II. Hunter syndrome is a rare disease caused by a deficiency of an enzyme necessary for the breakdown of sugars called glycosaminoglycans (GAGs). As a result of this enzyme deficiency, GAGs accumulate to toxic levels throughout the body, leading to progressive multiorgan disease.
Since 2006, the only FDA-approved treatment has been idursulfase (Elaprase) enzyme replacement therapy. Although Elaprase treats some of the somatic symptoms of Hunter syndrome, it does not treat neurological symptoms due to its inability to cross the blood-brain barrier. As a result, patients suffer progressive neurodegeneration and early death.
Since being diagnosed at age 2, my brother has gradually lost the ability to communicate verbally and is now entirely dependent on others for his care. It has been heartbreaking to see what this illness has taken from him and our family, and we constantly worry about what lies ahead.
The Hunter syndrome community is small, and in recent months several people – many of them teenagers and early adults – have died. I began to dread opening social media, where I often find news of yet another devastating loss. We desperately need better treatments that halt the physical and neurological decline of the disease, so families like mine can focus on living life to the fullest instead of worrying about how much time we have left.
That’s why the FDA’s recent approval of a new drug for Hunter syndrome should be considered a major achievement. On March 25, the FDA granted accelerated approval to Avlayah (tividenofusp alfa-eknm), a weekly enzyme replacement therapy designed by Denali Therapeutics to cross the blood-brain barrier and treat neurological and somatic symptoms. This approval represents a major breakthrough as the first new therapeutic innovation for Hunter syndrome in 20 years and the only FDA-approved therapy designed to treat the neurological symptoms of the disease.
Yet while Avlayah’s approval represents an important step, it remains far from a true victory for many families, including mine. Under the current FDA label, Avlayah is approved to treat only pediatric patients with Hunter syndrome up to 16 years of age.
The FDA’s choice to limit Avlayah to pediatric patients stems from the drug’s Phase 1/2 trial, which included patients ages 3 months to 13 years. However, this reflects the patients available for study during the trial, rather than a clinical determination that only pediatric patients would benefit from the treatment.
In fact, scientific evidence suggests that patients over 16 years of age would benefit significantly from Avlayah, as GAGs accumulate and cause disease progression in adults through the same pathophysiological mechanism observed in children. Because Avlayah reduces this accumulation of GAGs, its ability to treat disease progression should be the same in children and adults. This reasoning is consistent with previously approved enzyme replacement therapies for other diseases that do not have an upper age limit and is supported by the ongoing Phase 2/3 trial that includes participants up to 26 years of age, meaning Denali Therapeutics recognizes that the clinical benefit extends beyond the ages covered by the current FDA-approved label.
Therefore, even though accelerated approval brings therapies to patients more quickly, families like mine need the FDA to approve therapies based on what the science strongly suggests: adults with Hunter syndrome may benefit from Avlayah because of its ability to prevent disease progression and should have equal access to treatment.
Due to age restrictions set by the FDA, doctors must prescribe Avlayah off-label to help anyone over 16 years old. Insurance companies may therefore deny coverage, forcing patients, families, and providers to engage in a lengthy appeal process requiring multiple letters, calls, and meetings with no guarantee of success. During this time-consuming process, patients may experience further physical and neurological decline that can lead to irreversible organ damage.
In addition to harming patients’ health, this lengthy appeal process also puts considerable pressure on doctors. According to a 2024 survey from the American Medical Association, physicians and their staff spend an average of 13 hours per week writing prior authorization and appeal letters, leading about 90% of physicians to report increased burnout from this appeals process alone. If insurance coverage is ultimately denied, patients could face a staggering annual cost of between $270,000 and $811,000, depending on the dose required by their weight. By limiting Avlayah to pediatric patients, the FDA is creating harmful barriers to care that result in significant health and financial disparities.
Notably, some adults with Hunter syndrome can access Avlayah if they began treatment with the drug as part of a clinical trial when they were children. Although this continuity of treatment is necessary, it raises ethical concerns about equity of care and justice because patients with the same disease are treated differently depending on whether or not they had the opportunity and resources to participate in a clinical trial as children. Adults who did not participate in the first trials are forced to fight for access, leading to new health and economic disparities within the same patient community.
While Denali is conducting a phase 2/3 trial to confirm clinical benefit in a larger cohort of patients, including participants under the age of 26, these confirmatory trials may take years. Patients with Hunter syndrome do not have this wait time because disease progression is an ongoing process that can lead to irreversible organ damage. Additionally, there is no guarantee that the FDA will expand the label to include patients of all ages once the trial is completed. Because scientific evidence suggests adults will benefit from the treatment and similar therapies are not age-restricted, all adults with Hunter syndrome should have access to Avlayah now without having to wait for additional trials.
Thanks to advances in medicine, people with Hunter syndrome and other rare diseases are increasingly surviving into adulthood. While this is a remarkable achievement, rare disease clinical trials, policy discussions, and advocacy initiatives are still primarily focused on pediatric populations, leaving many adults overlooked. To improve outcomes, we must design clinical trials that recruit patients across the lifespan, ensure that patients are not denied therapies solely because of their age, and include adults in all education, policy, and drug development decisions regarding rare diseases.
Surviving into adulthood should not mean being excluded from hopeful therapies. Like so many others, my brother doesn’t have decades to wait for additional clinical trials with the potential for an expanded FDA label.
Nathan Grant is a medical/MBA student at Harvard Medical School and Harvard Business School, a researcher at Boston Children’s Hospital, a board member of Project Alive, and president and founder of Siblings with a Mission.
PakarPBN
A Private Blog Network (PBN) is a collection of websites that are controlled by a single individual or organization and used primarily to build backlinks to a “money site” in order to influence its ranking in search engines such as Google. The core idea behind a PBN is based on the importance of backlinks in Google’s ranking algorithm. Since Google views backlinks as signals of authority and trust, some website owners attempt to artificially create these signals through a controlled network of sites.
In a typical PBN setup, the owner acquires expired or aged domains that already have existing authority, backlinks, and history. These domains are rebuilt with new content and hosted separately, often using different IP addresses, hosting providers, themes, and ownership details to make them appear unrelated. Within the content published on these sites, links are strategically placed that point to the main website the owner wants to rank higher. By doing this, the owner attempts to pass link equity (also known as “link juice”) from the PBN sites to the target website.
The purpose of a PBN is to give the impression that the target website is naturally earning links from multiple independent sources. If done effectively, this can temporarily improve keyword rankings, increase organic visibility, and drive more traffic from search results.