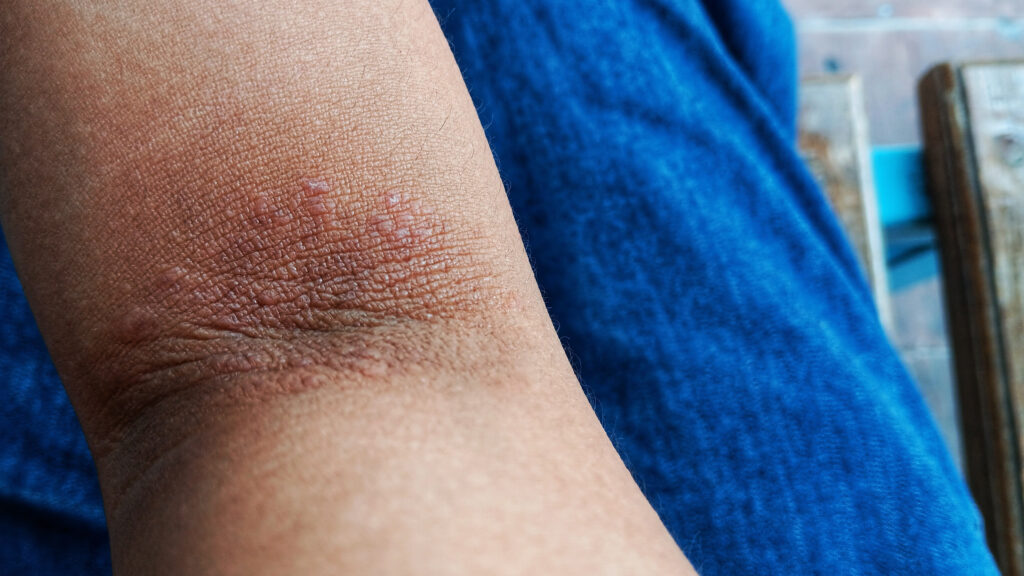
Eczema drug Kymera succeeds in first trial, could rival Dupixent
Kymera Therapeutics announced Monday that, for the second time this year, it had successfully completed an early trial of a closely watched drug intended to rival Sanofi and Regeneron’s blockbuster treatment Dupixent.
The phase 1 BroADen study enrolled 22 patients with moderate to severe eczema (atopic dermatitis), all of whom took Kymera’s investigational pill, KT-621. After four weeks of treatment and two weeks of follow-up, their overall disease severity dropped by 63% on average, according to the commonly used Eczema Area and Severity Index (EASI) score. This was a slightly better improvement than Wall Street had hoped for, according to a note announcing the results from Leerink Partners.
Watertown, Mass.-based Kymera, which has a market value of about $4.8 billion, is seeking to compete with Dupixent, a treatment developed by Sanofi and Regeneron that generated more than $14 billion in revenue last year and has been approved for the treatment of eczema, asthma and chronic obstructive pulmonary disease, among other conditions. The blockbuster drug is an antibody that blocks IL-4 and IL-13, immune signals called cytokines that can cause inflammation.
This article is reserved for STAT+ subscribers
Unlock this article, plus in-depth analysis, newsletters, premium events and news alerts.
Already have an account? Log in
See all packages
Firm Law
Agen Togel Terpercaya
Bandar Togel
Sabung Ayam Online
Berita Terkini
Artikel Terbaru
Berita Terbaru
Penerbangan
Berita Politik
Berita Politik
Software
Software Download
Download Aplikasi
Berita Terkini
News
Jasa PBN
Jasa Artikel



